Oxygen - gas without color and a smell which is a part of air. It is necessary for breath and burning and is some their most widespread elements on Earth.
Instruction
1. Oxygen is chemical element 7A of group of a periodic system of elements. This element belongs to family of chalcogens. Depending on its aggregate state, its properties strongly change, and the crystal lattice is subject to change.
2. First of all it should be noted that crystal lattices are characteristic only of solid substances. Therefore if to speak about that at oxygen, then it should be taken into account under what conditions oxygen can be in such aggregate state.
3. It is known that under usual conditions oxygen is in gaseous state, at decrease in temperature to - 194 wasps it turns into liquid of blue color and only at a temperature - 218.8 wasps take a form of snegoobrazny weight with crystals of blue color.
4. Except rather low temperature, the firm condition of oxygen requires the huge pressure which exceeds atmospheric approximately in 52 thousand times (these are about 5.4 gigapascals).
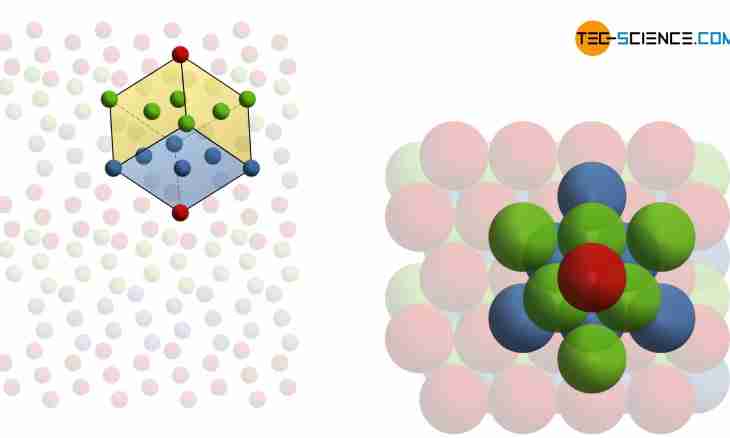
5. At further increase in pressure, solid oxygen becomes red. In such state it has a molecular crystal lattice. Contrary to expectations of scientists who considered that atoms in knots of a molecule will be located in the form of a ring as at sulfur atoms, atoms of oxygen are located absolutely differently.
6. Atoms of oxygen gather groups on eight atoms, but at the same time they do not form a ring, and are located in the form of a rhombohedron. This figure represents the flattened cube. Thus, the molecule consisting of eight atoms turns out. It has molecular formula O8.
7. In a molecule, atoms are connected by very strong covalent links, but because of a weak intermolecular attraction, all substances with molecular crystal lattices have the small firmness, high volatility and low temperature of melting.
8. Solid oxygen exists in several crystal allotropic modifications. The steadiest alpha form which has a body-centered rhombic crystal lattice. Less steady - a beta form with a hexagene crystal lattice. Also there is a g-form, it is most of all similar to an alpha form on the properties, but type of a crystal lattice at it cubic.
9. Unfortunately, solid oxygen is extremely unstable therefore it did not find any practical application. At the slightest pressure decrease its crystal lattice collapses and it evaporates.