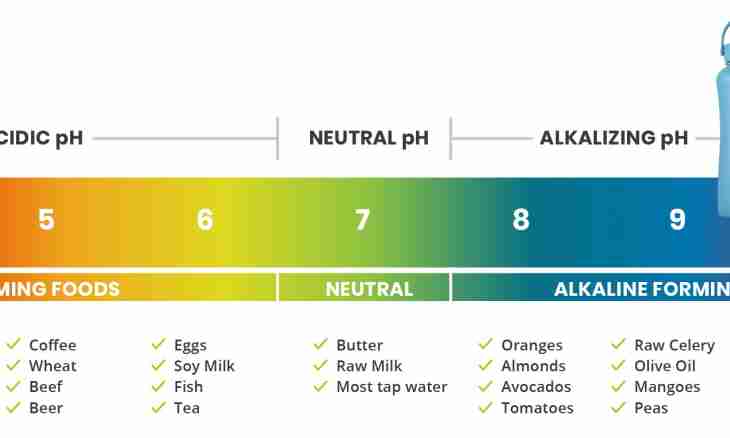
In chemistry exist alkaline, acidic and neutral environment. They have qualitative difference which consists in a pH value of pH (from Latin pundus hydrogenium - "hydrogen weight").
PH value of the environment
In advertizing often slips a concept of a pH value of the environment. Consumers are assured that it remains at the normal level, and the products of firm are safe for people.
Usual water at the room temperature always contains a small amount of positively charged cations of hydrogen and also negative hydroxide-anions. They are formed as a result of reversible dissociation. One liter of water without impurity contains 10*7 mol of cations of hydrogen and as much anions. For convenience of designation entered a concept of a pH value which for clear water is equal to 7. This substance is neutral by the nature. There are also other neutral environments.
At acids and alkalis the pH value accepts other values. In a case with acids their dissociation in water can be reversible and irreversible. Anyway the maintenance of cations of hydrogen in such environment decreases. Irreversible dissociation is characteristic of strong acids, for example, salt. Its solution contains 10*2 mol of cations of hydrogen, pH of such solution is equal to 2. Apparently, the exponent defines value of an indicator of the environment. It is the logarithm of quantity of cations taken with the return sign. For acids it always less than 7. The acid is stronger, the pH is lower. With alkalis the situation is a little differently. At their dissociation in water the excess quantity negatively charged hydroxide-anions appears. They take a part of cations of hydrogen and thus reduce their quantity. It becomes less than 10*7 mol. And in this case the value of an indicator is equivalent to an exponent. Strong alkalis dissociate is irreversible and their pH varies from 7 to 9. Weak alkalis which dissociation is reversible process have pH values from 9 and above.
Indicators
It is possible to define type of any liquid environment by special substances. These substances are called indicators. They are capable to change color depending on Wednesday where they are placed. Carry phenolphthalein and litmus to them. In the neutral environment all indicators do not change the color. Initially violet litmus placed in acid solution gets a bright red shade and becomes blue in alkali. Colourless phenolphthalein as the indicator is applied not so widely as it equally reacts to the silnoshchelochny and neutral environment. But it well shows a lack of cations of hydrogen (acidity of the environment), being painted in red.